|
Daily Science: Students where given simple chemical equations to balance.
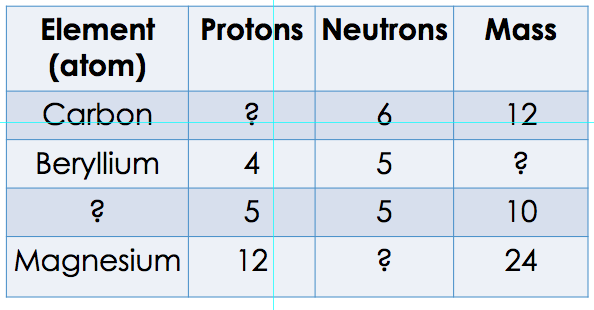
We spent the rest of the hour working on balancing chemical equations of a handout. Students worked on their own, and several equations were also presented on the board. Students should complete handout for homework. Students are to show their work (attached on loose leaf) Due Friday. Test on Essential Elements is scheduled for this Thursday. Students will be able to use their essential elements chart during the test. The test will cover the following: Chapter 3 lesson 1: Students should know The difference between an element and a compound and be able to include and example of each. The important groups of organic compounds, what elements they are made off and where they are found in a cell. Parts of the atom (protons, neutrons electrons; where they are found and their charges). Basic use of the periodic table. What information can we get from the periodic table for each element? ( What is the atomic number and what is it equal to? what is the atomic mass, and what is it equal to? How can we calculate the number of neutrons?) Balancing of simple chemical equations. The students should also know the % of the most important elements. E.g. which one is the most abundant element, second most abundant, third etc (see the pie chart we drew in class).
0 Comments
Daily Science: From your research on the essential elements, which element is considered a building block upon which all other elements depend on? What would happen if we were deficient of this element?
Today in daily science we completed the essential elements chart and discussed the importance of each element, the effects of deficiency and the food sources of each element. The students also started working on their Wanted Element Poster. Students are working in groups of two for this poster. Tomorrow students will be given time during class to work on their projects. After that they are on their own and they will have to complete the rest of the work at home. Poster is due on Tuesday September 20th. Please consult the guidelines in the previous post for this project. . Daily Science:
Use the periodic table to name the following elements: 1. What element contains 20 protons? 2. What element contains 11 electrons? 3. What element contains 6 neutrons? 4. What element has the atomic number 8? 5. What is the atomic mass of Nitrogen? Start thinking about which element you would like to research for your Essential Element Wanted Poster. Please read below for the requirements: WANTED POSTER REQUIREMENTS 1. Construct a poster which displays as much information about your assigned element as possible. Be sure to include a title and name. 2. Your poster should be in the form of a police "wanted" poster 3. Your poster should be on paper. 4 Include at least one large illustration that is related to your topic. You can make your own drawings or use pictures or photocopies from magazines or books. 5. All information on your poster must be written in your own words and factually correct. 6. Neatness counts! Your lettering must be neat and straight. Do not have cross-outs, smudges, stains, wrinkles, or stray marks on your poster. 7. Spelling and grammar count too! 8. Use your imagination. Make an interesting and exciting poster! ITEMS THAT MAY BE INCLUDED ON YOUR ELEMENT POSTER: 1. WANTED FOR: These are particular "crimes" or uses of your element. Why is it important? 2. ALIAS OR AKA'S (Also Known As): Other names used for your element. Include its chemical symbol, historical names, common names, slang terms, etc 3. DESCRIPTION: Color, weight (atomic mass), distinguishing characteristics (atomic number and structure, metal or non-metal, other physical properties), density, melting point, luster (shiny or dull), hardness (moh's scale) etc. 4. MODUS OPERANDI: (Mode of Operation): How your element behaves and reacts (chemical properties). Reactive or nonreactive, poisonous, flammable, corrosive, explosive, etc. 5. FIRST ARRESTING OFFICER: The name and information about the person who discovered your element, if available. 6. ARREST REPORT ON FIRST ARREST: a) Date arrested: When was it discovered? b) Particulars: How was it discovered? 7. PRIORS: What has it been used for in the past? 8. LAST KNOWN WHEREABOUTS: Location where your element is found; its abundance on Earth 9. KNOWN ASSOCIATES: List other elements with which your element is often associated or combined with in ores and compounds. 10. WARNING: Is it armed and dangerous, or good natured and well behaved? EVALUATION CRITERIA [3] Correct size: on printer paper [2] Includes name and title [5] Neat; no smudges, stains, wrinkles, or marks; not torn [5] Straight, easy-to-read lettering [5] Correct spelling and grammar [5] Colorful, attractive, and eye catching [5] Follows "wanted poster" format [15] Clear, neat illustration(s) related to topic [15] Informative [10] Written in own words and easy to understand [15] Creative [15] Information is factually correct Poster should be unique. No copy work. Total Score: 100 Daily Science Question: What is the difference between an element and a compound?
Homework: Chapter 3 vocabulary (assigned on Tuesday) is due tomorrow Friday. Review for the test for 5 min
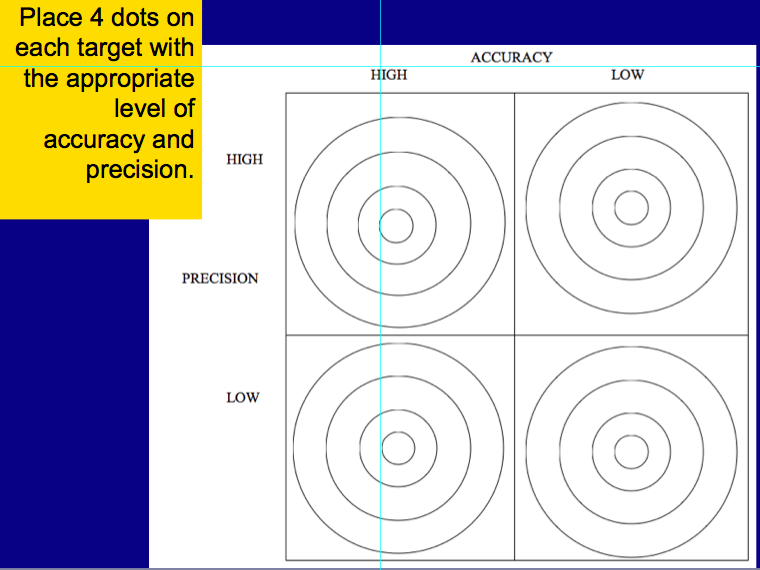
After the test please read and answer the questions on the handout about accuracy and precision. We will discuss it in class tomorrow. Also begin Chapter 3 vocabulary which is due Friday Sept. 9th, Daily Science: Convert the following:
1.5 L = _______ mL 2.198 g = _______ kg 3.75 mL = _____ L 4.120 mg = _____ g 5.2500 m = _______ km Homework: Review Length , Mass and volume measurement for test on Tuesday. Daily Science:
Convert the following: 1. 1000 mg = _______ g 2. 1 L = _______ mL 3. 14 km = _______ m 4. 109 g = _______ kg 5. 160 cm = _______ mm 6. 250 m = _______ km Homework: Complete chapter 2 vocabulary Start preparing handwritten page of notes that can help you during the measurement test on Tuesday. |
About the BlogDaily classwork and homework updates. Archives
April 2017
|
 RSS Feed
RSS Feed

